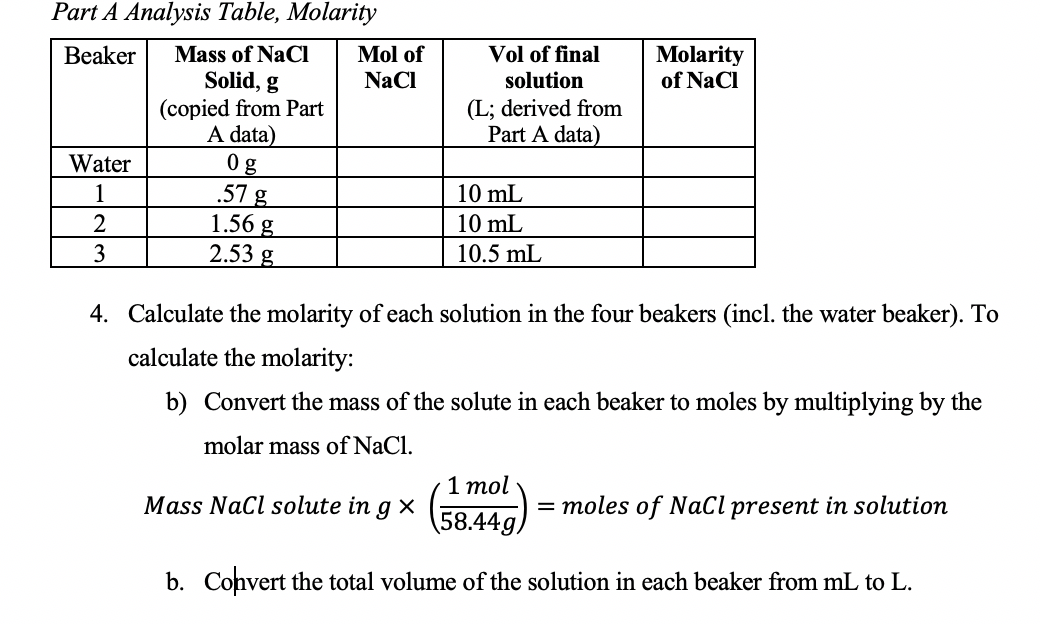
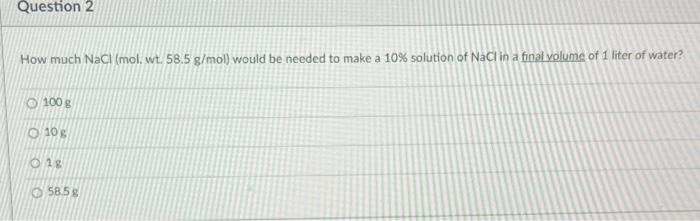
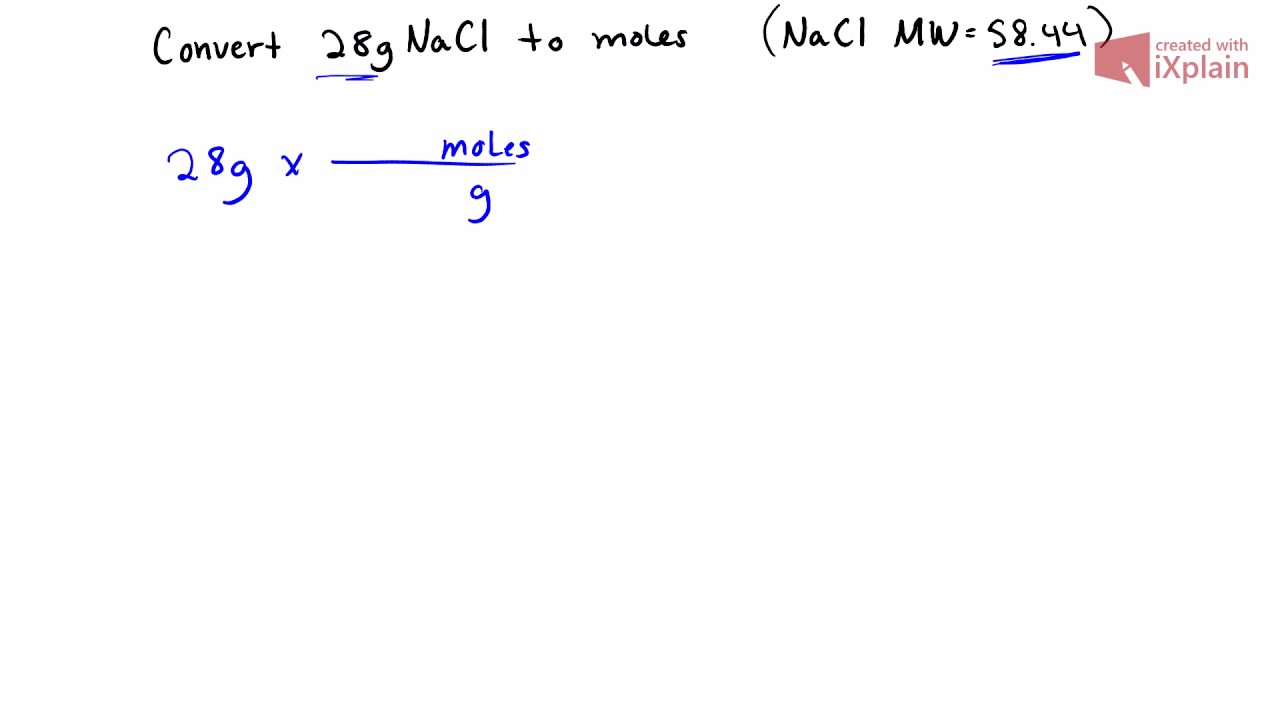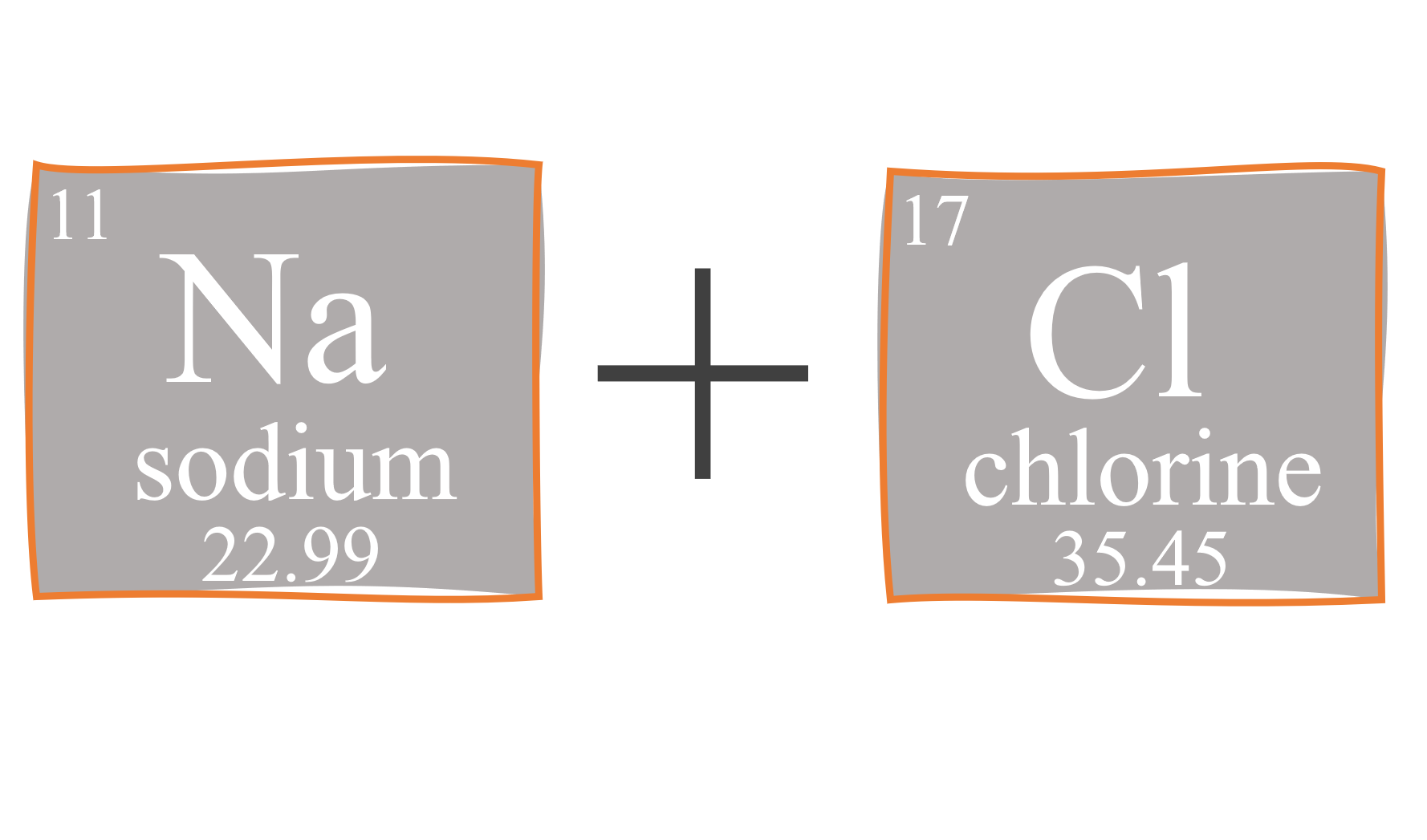SOLVED: 7J Iinul Convert the mass of sodium chloride collected to moles. Moln Gci 66 ,44 3/mol 3089x Inul 0,00354(Mul 58 449 'Mul Divide both of your results from the preceding two

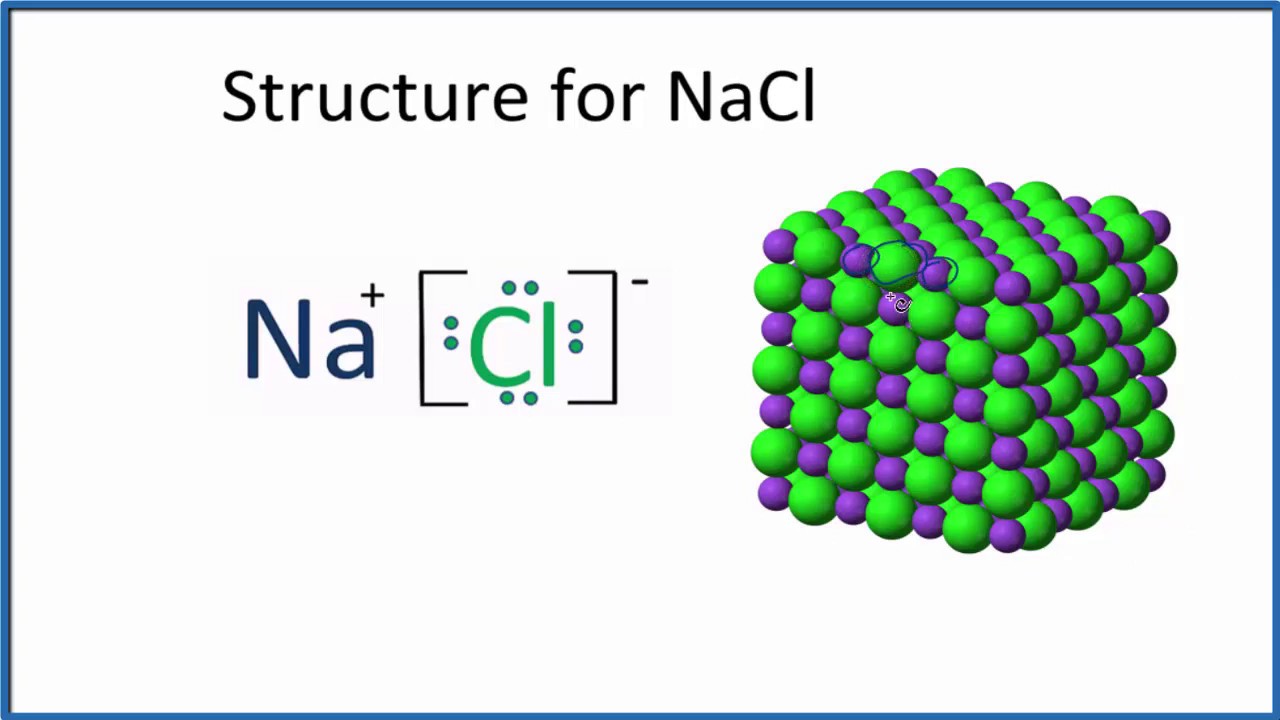
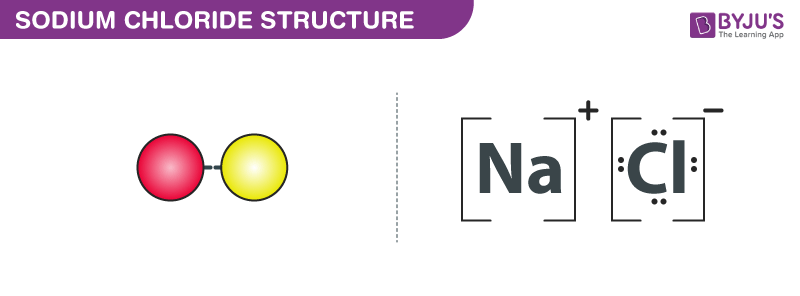
Formula mass of NaCl is 58.45 g mol ^-1 and density of its pure form is 2.167 g cm ^-3 . The average distance between adjacent sodium and chloride ions in the