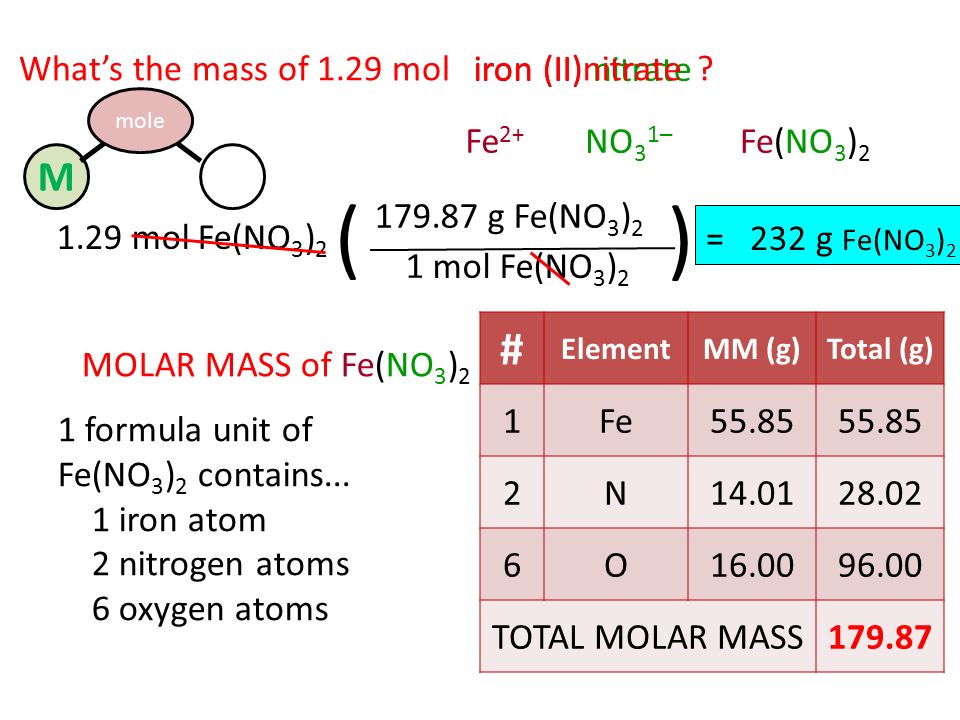
Iron is produced by the reduction of iron (III) oxide using carbon monoxide. Fe2O3(s) + 3CO(g) 2Fe(s) + 3CO2(g). How much Fe is produced from 1 kg of Fe2O3? - Quora
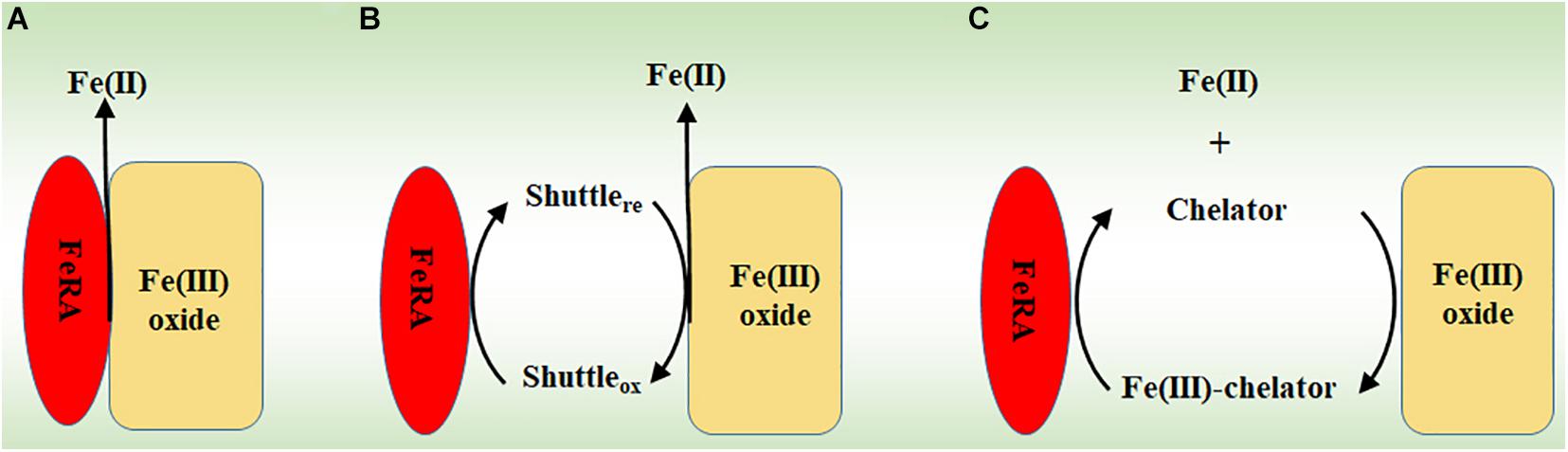
Frontiers | The Proposed Molecular Mechanisms Used by Archaea for Fe(III) Reduction and Fe(II) Oxidation

Schematic molecular orbital (MO) diagrams of 1 Fe(CO) 4 (left) and 3... | Download Scientific Diagram

If 3.50 mol of Fe reacts with 3.00 mol of oxygen in the following reaction: 4 Fe(s) + 3 O2(g) → 2 - Brainly.com

Characteristics of Fe and Mn bearing precipitates generated by Fe(II) and Mn(II) co-oxidation with O2, MnO4 and HOCl in the presence of groundwater ions - ScienceDirect