
Johnson & Johnson Announces European Commission Approval for Janssen's Preventive Ebola Vaccine | Johnson & Johnson
Johnson & Johnson (Janssen Pharmaceutical Cos.) - Single-Shot COVID-19 Vaccine (formerly JNJ-78436735, Ad26.COV2.S)

Johnson & Johnson Announces Donation of up to 500,000 Regimens of Janssen's Investigational Ebola Vaccine to Support Outbreak Response in Democratic Republic of the Congo (DRC)

J&J's Heaton becomes face of COVID-19 booster debate in first months as Janssen vaccine chief | Fierce Biotech
UK, Baptist Health Lexington and Norton Healthcare Begin Recruiting Participants for Phase 3 COVID-19 Investigational Vaccine Clinical Trial | UKNow
AdventHealth begins recruiting participants for Phase 3 COVID-19 Investigational vaccine clinical trial | AdventHealth Research Institute
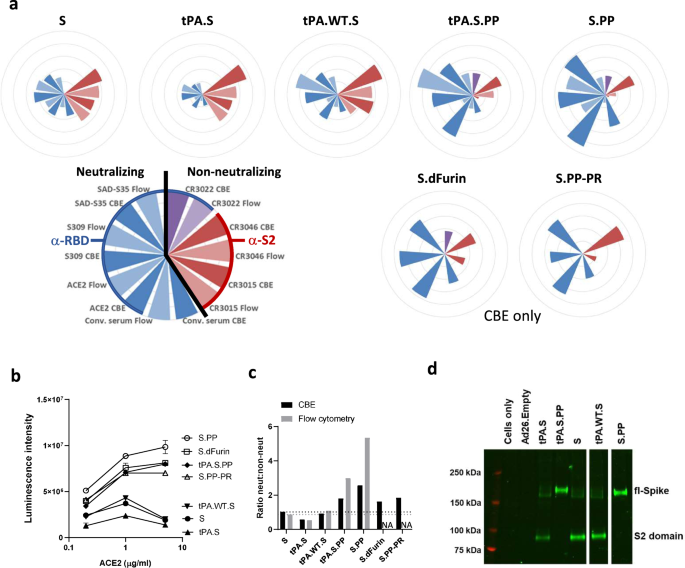
Ad26 vector-based COVID-19 vaccine encoding a prefusion-stabilized SARS-CoV-2 Spike immunogen induces potent humoral and cellular immune responses | npj Vaccines
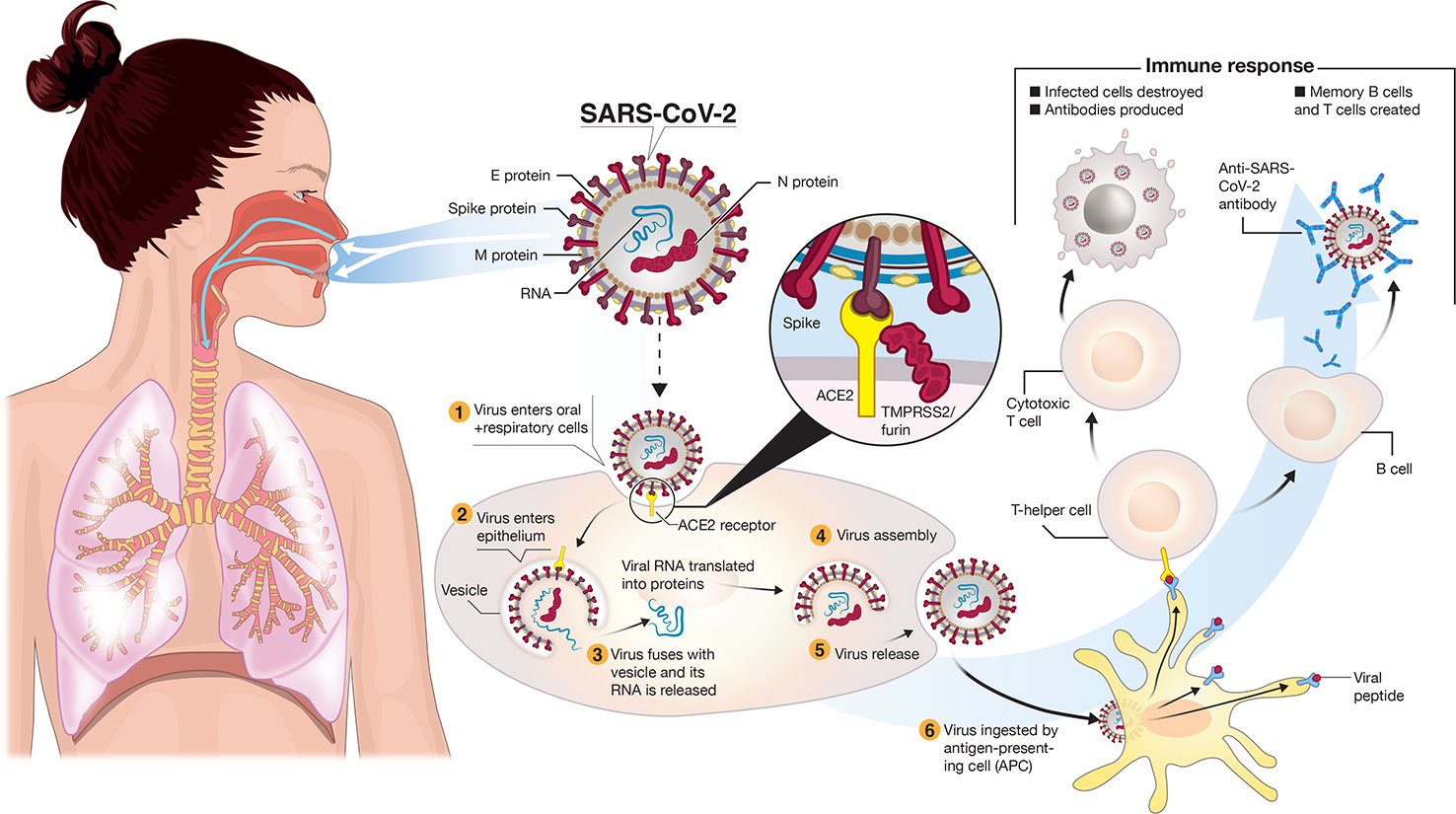
Frontiers | A Snapshot of the Global Race for Vaccines Targeting SARS-CoV-2 and the COVID-19 Pandemic





.png)


